Discussion
Discussion
Rett syndrome treatment
Last updated Dec. 18, 2025, by Marisa Wexler, MS

Rett syndrome is a genetic disorder marked by developmental abnormalities, motor impairment, and communication difficulties.
There is no cure for Rett syndrome, and no treatment addresses the root cause of the disease. However, therapies are available to help with managing Rett syndrome symptoms and improving quality of life for patients and their families. Research to find new treatments is ongoing.
Most people with Rett will require supportive care throughout their entire lives, though specific types of care needed may vary over time. When children are diagnosed, their families are advised to talk with healthcare providers to develop a personalized Rett syndrome care plan and to maintain open communication about therapy options that may be helpful as their situation evolves.
Rett syndrome support for caregivers is also a crucial part of caring for families affected by the disease.
Medical treatments
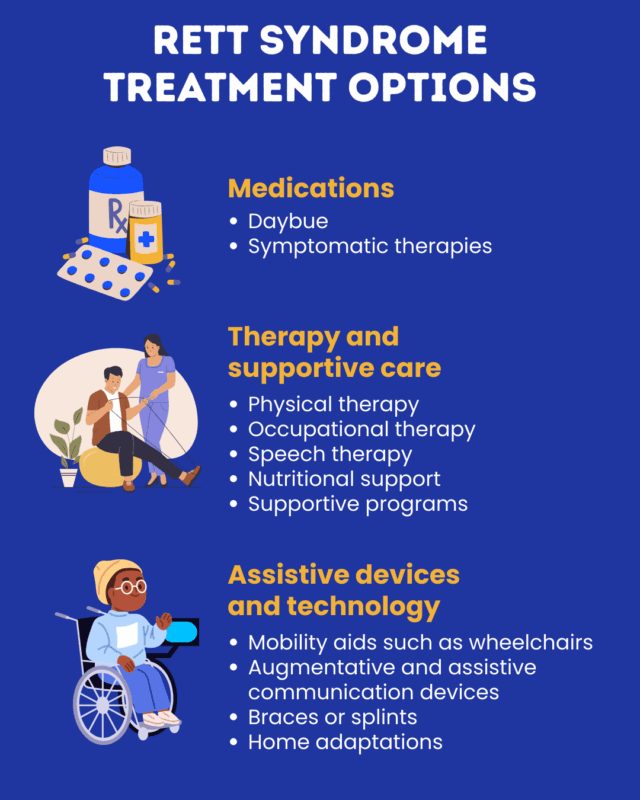
Two formulations of trofinetide, Daybue and Daybue Stix, sold by Acadia Pharmaceuticals, are approved in the U.S. for Rett syndrome. Other medications are not specifically Rett syndrome treatments, but nonetheless may be used to help manage its symptoms.
Approved treatments: Daybue and Daybue Stix
Daybue became the first Rett syndrome medication approved by the U.S. Food and Drug Administration in 2023 and by Health Canada a year later, and it is currently under review in Europe. It is indicated for Rett patients 2 years and older.
The treatment is broadly thought to help reduce brain inflammation and improve the health of connections between nerve cells. A Phase 3 clinical trial showed Daybue was significantly better than a placebo at improving scores on a caregiver-rated assessment of Rett symptoms, with long-term data suggesting continual improvements after several years of treatment.
No serious side effects were reported in clinical trials of Daybue, though the therapy is known to frequently cause gastrointestinal side effects such as diarrhea and vomiting.
Daybue comes as a ready-to-use liquid. A dye- and preservative-free powder formulation, called Daybue Stix, which must be dissolved in liquid before administration, was approved in the U.S. in late 2025.
Medicines for symptom management
Apart from Daybue and Daybue Stix, a wide range of medications may be used to manage Rett symptoms. Generally, this involves medications that are approved to help relieve specific issues in the general population. Rett syndrome symptom management may include treatments to:
- reduce seizures, such as antiepileptic drugs
- ease digestive issues, such as laxatives for constipation or acid reducers for reflux
- normalize heart rhythms and help with respiratory issues, such as Rett syndrome breathing treatments
- improve mobility, such as botulinum toxin injections to relieve muscle spasticity
- help promote sleep, such as melatonin
- treat mental health issues, such as antidepressants and drugs for anxiety.
Therapies and supportive care
Apart from medications, Rett syndrome therapy options and supportive care may be used to help improve the health and well-being of patients.
- Physical therapy for Rett syndrome and other disorders or for people with injuries, also called physiotherapy, focuses on exercises and stretching to maximize physical function. Physical therapists can also work with patients and families on the use of Rett syndrome assistive devices such as braces and wheelchairs.
- Occupational therapy for Rett syndrome patients involves a therapist who works with the patient and their family to design strategies that will make it easier to navigate day-to-day life, such as finding easier ways to go about dressing or feeding.
- Speech and language therapy can help patients better communicate and navigate social interaction, whether by speaking or using assistive devices. Speech therapy can help individuals with issues swallowing.
- Nutritional support can help ensure that patients are getting all the nutrients they need to stay healthy, and also help manage digestive symptoms and minimize the risk of vomiting and aspiration. Nutritional support may involve administering supplements of specific vitamins, adjusting the diet, or feeding through a tube inserted surgically into the stomach (gastrostomy).
- Supportive care like one-on-one aids and adaptive programs can help people with Rett successfully attend school, engage in the workforce, and participate in sports and other social activities. It’s recommended that families should reach out to schools and other local resources to learn about support that’s available in their community.
- Surgery may be recommended in some cases to manage issues like scoliosis, which refers to a sideways curvature of the spine.
- Other types of Rett syndrome rehabilitation have been explored, such as hippotherapy, massage therapy, music therapy, and hydrotherapy where exercises are done in water. There isn’t much data on the effects of these types of therapy for people with Rett syndrome, but it’s generally thought that they may help patients build and maintain functional skills, as well as provide outlets for social interaction and emotional connection.
Assistive devices and technology
Most people living with Rett syndrome will rely on assistive devices and technology to help manage disease symptoms and daily life. Some types of technology that are commonly used by people with Rett include:
- mobility aids such as wheelchairs
- augmentative and assistive communication devices that can help patients make themselves better understood. Tools for Rett syndrome non-verbal communication can range from cardboards with pictures that patients can point at, to high-tech devices that track eye movements.
- braces or splints to help reduce the risk of scoliosis and joint problems
- home adaptations to make day-to-day activities easier, such as lifts, chairs or bars in the shower, bedside toilets, and modified hygiene tools.
Since every family affected by Rett syndrome is unique, not every type of adaptive aid is suitable for every patient. Families are encouraged to work with their clinical care team to identify the equipment that will be helpful and that can be used safely and comfortably. It’s also important to consider financial and logistical issues. For example, if a child with Rett syndrome uses a wheelchair, it may be necessary for the family to get a wheelchair-accessible vehicle.
Experimental and emerging treatments
Rett syndrome therapy research is ongoing. Clinical trials are carefully-designed studies in humans that are used to test the safety and effectiveness of potential new treatments. Experimental treatments that are being tested in Rett syndrome clinical trials include:
- Anavex 2-73 (blarcamesine)
- NA-921 (bionetide)
- AMO-04 (tianeptine)
- gene therapies and gene-editing therapies.
Anavex 2-73 is an oral therapy that Anavex Life Sciences is developing for Rett syndrome. A Phase 3 trial in adults with Rett syndrome showed the therapy was significantly better than a placebo at easing Rett symptoms. A Phase 3 study in children with Rett syndrome also showed a tendency toward symptom easing with the therapy, but the difference from a placebo was not statistically significant, missing that study’s main goal.
NA-921 is a therapy given orally or via a feeding tube to promote brain cell health that Biomed Industries is developing for Rett syndrome. A Phase 2/3 trial suggested the therapy may help normalize behaviors in Rett patients, and a larger Phase 3 study is ongoing.
AMO-04 is an antidepressant therapy that isn’t approved in the U.S., but is used to treat mental health disorders in some other countries. Amo Pharma and Numedicus are collaborating to develop this therapy for Rett syndrome; it is currently in Phase 2 clinical testing.
Nearly all cases of Rett syndrome are caused by mutations in the gene MECP2. Gene therapies such as TSHA-102 and NGN-401 aim to deliver a healthy copy of the faulty gene to the body’s cells, while gene-editing therapies remove, add, or alter sections of DNA. In either case, the ultimate goal is to address the underlying genetic deficit that causes Rett syndrome. Multiple gene therapies and gene-editing treatments for Rett syndrome are currently in clinical testing.
Rett Syndrome News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
Recent Posts
- The stranger on the beach who performed an act of real charity
- Imagining the moments my late daughter would now be experiencing
- Rett mutations may disrupt DNA-packaging proteins in nerve cells
- Lamenting a broken memento of a big night out
- Study lays groundwork for mutation-specific Rett treatments
- The one question I wish my nonverbal child could answer
- The most difficult conversation I have ever had
- Experts recommend Daybue as first-line therapy for Rett syndrome
- Guest Voice: Like stars, the grief of losing my sister is always present
- The spa, the snow, and the Make-A-Wish gift that keeps giving
Related articles
-
 Discussion
Discussion